Attractive and safe chemical experiments linked to educational programmes
Chemiluminescence
Chemiluminescence is the emission of cold light as a result of a chemical reaction. In a chemiluminescent reaction, reactive intermediates are formed which enter electronically excited states. Subsequent transition back to ground state is accompanied by a release of energy in form of light. The mechanism is depicted in Fig. 5.

Compounds A and B react together forming C* and D while C* is in electronically excited or energetically rich state. C* can get rid of its extra energy by several pathways:
- Non-productive non-radiative transitions producing heat (undesirable process)
- Direct deexcitation of C* by a radiative transition with concomitant emission of light characteristic for C*
- Indirect deexcitation – C* transfers its extra energy to a a suitable fluorescer that can act as a temporary energy acceptor and then emit the energy in form of light. The emission spectrum is then dependent on the structure of fluorescer. This can be used with advantage since the colour of the emitted light can be easily varied.
As was mentioned in the beginning, the best-known and first well-documented example of deliberately induced, man-made chemiluminescence was the discovery of elemental phosphorus by a German alchemist H. Brand (Fig. 6). The waxy white phosphorus is slowly oxidized at room temperature giving off faint greenish glow. The formation of excited reactive intermediate (PO)2 and HPO is responsible for the green glow.

The term “phosphorescence” is often wrongly associated with phosphorus oxidation. Phosphorescence, like its cousin fluorescence, is a strictly physical phenomenon.
Today´s chemistry offers a plethora of chemical reactions that are known to give chemiluminescence. One of the traditional examples is oxidation of luminol with hydrogen peroxide, oxidation of siloxenes, reduction of RuIII complexes by tetrahydridoborate anion, oxidation of lophine, pyrogallol oxidation in the presence of formaldehyde (Trautz-Schorigin reaction), chemiluminescence of singlet oxygen, but arguably the most widespread are activated phenolic oxalates whose chemistry was mostly researched by American chemist M. Rauhut in the sixties and seventies. Chemiluminescence of these oxalates is used in commercial glow-sticks.
The success of the reaction of aryl oxalates with hydrogen peroxide (also called peroxyoxalate chemiluminescence) lies in its high efficiency, relatively low cost (in comparison with other systems), long reaction times and easy colour tunability by a judicious choice of fluorescer, spanning from blue to infrared. It was measured that up to 23% of the potential chemical energy of the peroxyoxalate system is converted into light which ranks it among the best performing non-enzymatic chemiluminescent reactions.
The best-known literature examples of phenolic oxalates include DNPO (bis-(2,4-dinitrophenyl)oxalate) and TCPO (bis-(2,4,6-trichlorophenyl)oxalate). However, the use of these esters for school demonstrations is rather unsuitable because during their reaction with hydrogen peroxide, highly toxic or unpleasantly smelling phenols are released (2,4-dinitrophenol and 2,4,6-trichlorophenol).
This kit contains a non-toxic alternative with a pleasant smell – bis-vanillyl oxalate (BVO), an ester of vanillin and oxalic acid that has a similar chemical behaviour but lacks these disadvantages.
The generally accepted mechanism of this reaction is described in Scheme 1:
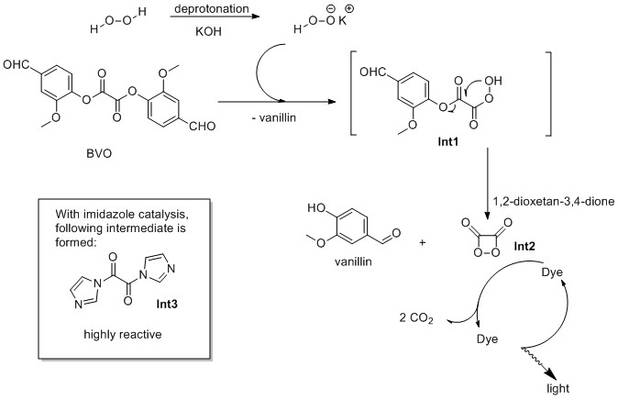
Hydrogen peroxide is deprotonated by a base to afford hydroperoxide anion which shows higher nucleophilicity that the parent hydrogen peroxide. This anion reacts then with BVO in the first step while 1 molecule of vanillin is released and the half-reacted intermediate Int1 is formed. Int2 then undergoes an intramolecular cyclisation to give off another moleculele of vanillin and a strained four-membered endoperoxide heterocycle 1,2-dioxetane-3,4-dione (Int2). This compound is energetically rich and highly unstable, and transfers its energy to a molecule of a fluorescent dye (fluorescer) which in turn emits light. The complex mechanism of energy transfer is accompanied by formation of 2 molecules of CO2 from Int2.
Apart from bases like KOH (or sodium acetate, sodium salicylate, etc.), the reaction can be also catalyzed by nucleophilic acylation catalysts, for example by imidazol. The catalytic effect of imidazol is caused by its ability to convert BVO to a much more reactive bis-imidazolyl oxalate (intermediate Int3) which reacts considerably faster with hydrogen peroxide.
The peroxyoxalate chemiluminescence offers a lot of didactic possibilities. A broad selection of chemical laws and rules can be easily demonstrated with this reaction – for example phase transfer phenomena, dependence of reaction rate on stirring rate, temperature, presence of catalysts, inhibitors, etc.
Since scientists can measure even the tiniest amounts of light, they can use chemiluminescence to detect trace amounts of chemicals. Chemiluminescent reactions find therefore frequent use in analytical chemistry.
The peroxyoxalate chemiluminescence finds application in analytical chemistry for detection of low levels of hydrogen peroxide. Another example of a very sensitive analytical method is determination of nitric oxide concentration in air by a chemiluminescent reaction of nitric oxide with ozone. This is routinely applied for online-monitoring of NO concentration in the air.






